Buy Potassium Ferricyanide ACS (13746-66-2) MF: K3[Fe(CN)6] | MW: 329.24. Get High Quality Potassium Ferricyanide ACS (13746-66-2) from ChemieR.
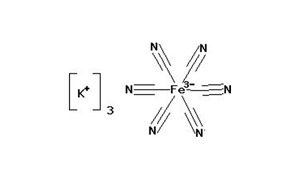
Potassium Ferricyanide ACS is a chemical compound and a bright red salt. It is highly soluble in water and appears as a yellow-green fluorescence. Potassium ferricyanide is the chemical compound with the formula K3[Fe(CN)6]. This bright red salt contains the octahedrally coordinated [Fe(CN)6]3− ion. It is Light sensitive. The ACS (American Chemical Society) grade denotes a high level of purity suitable for laboratory and analytical applications. Potassium Ferricyanide ACS is commonly known as potassium hexacyanoferrate(III) or red prussiate.
ChemieR offers a variety of high purity salts and solutions of alkali metals, alkaline earth compounds, perchlorates, heavy metals, rare earths, transition metals, silver/precious metals, and triflates. ChemieR covers wide range of inorganic chemicals, specializing in low moisture and low trace metal grade materials.
ChemieR is innovative and unique product range in laboratory chemicals which includes organic reagents, inorganic reagents, and solvents. Order from our ChemieR brand to meet your budget whilst not compromising on the quality.
In-addition ChemieR also distributes in numerous Laboratory Supplies, Chemicals, Equipment, Instruments, Reagents, Standard Solutions, Buffers, Biological Stains & Indicators and many more, for more information please browse our website (https://chemiereagents.com) or email us sales@chemiereagents.com we will be happy to help you. All ChemieR Products are exclusively distributed by Dawn Scientific Inc (https://dawnscientific.com)
Application :
- Potassium Ferricyanide ACS can be used as a mild reducing agent in certain chemical reactions.
- Potassium Ferricyanide ACS is commonly used as a reagent in analytical chemistry, especially in the determination of reducing sugars.
- Potassium Ferricyanide ACS finds applications as a reagent in various laboratory procedures, including the detection of metal ions and as an oxidizing agent in organic synthesis.
Benefits :
- Meet the toughest regulatory standards for quality and purity
- Used in oxidation-reduction reactions due to its ability to donate electrons
- Relatively non-toxic in nature






Reviews
There are no reviews yet.